Today’s the day: AICA — the Audit Intelligence Compliance Assistant — is officially launched and available to the life sciences industry. The website is live at aica.thefdagroup.com, and we’ve published our full press release here.
If you read last week’s Insider sneak peek email, you already have a strong foundation on what AICA is and why we built it. A lot of what follows will be familiar.
But today, with the platform officially in the market, we’re going deeper — more detail on the technology, the partnership, the security architecture, the implementation process, and what comes next. Consider this the definitive reference for everything AICA.
In case your time is short: AICA is an AI-powered compliance auditing platform purpose-built for pharmaceutical and biologic companies. You upload your QMS documentation, select which FDA regulations to audit against, and AICA analyzes all of it — not a sample — identifying gaps between what your documents say and what regulations require. You get results in hours instead of days or weeks. Every finding is reviewed by your quality team before the final report is generated. We built it in partnership with EPAM Systems, and it’s trained by 15+ experienced FDA and ISO auditors on real-world pharma QMS documentation.
If you’re brand new to AICA, don’t worry. We’re starting from the top!
The problem we built AICA to solve
If you run a quality or regulatory affairs program, you probably know this tension well: you need to run audits, but they’re slow, resource-intensive, and often limited in scope relative to your full QMS.
We studied this problem more closely by running an internal benchmarking study where three of our experienced auditors independently reviewed the same set of QMS documentation against the same regulations.
The overlap in their findings (the observations they agreed on) ranged from just 1.2% to 14%. Again, these were three extremely qualified auditing professionals with the same documents and regulations, and their results barely overlapped.
That’s not a knock on those auditors; it’s the nature of compliance reviews that are totally manual. Human audits are inherently subjective. Different auditors notice different things, prioritize different risks, and interpret requirements through the lens of their own experience.
On top of that, we found that only about 30% of auditable documentation actually gets audited in a given cycle due to time, budget, and human bandwidth constraints. The rest goes unexamined until the next audit — or until an investigator finds something first.
Quality leaders have faced the same frustrating choice for decades: conduct thorough compliance audits that take their teams off critical work for weeks, or use sampling methods that leave most of their QMS unexamined.
We built AICA to give them a third option.
What AICA actually does
Put simply, AICA is an AI tool that audits your QMS documentation against FDA regulations.
You upload your policies, procedures, and work instructions.
You select which regulations you want to audit against.
AICA analyzes everything you upload — not a sample, all of it — and identifies gaps between what your documents say and what the regulations require.
You get results in hours, not the days or weeks it takes a traditional audit.
In our internal testing, AICA is achieving roughly 50% overlap with the combined findings of all three human auditors — meaning it consistently captured a substantial share of what multiple experts found, across every document, every time. And it does so across 100% of your documentation, not just the ~30% that typically gets reviewed.
To be clear, AICA doesn’t replace auditors. It gives them a baseline across your entire QMS, so they can focus their expertise where it matters most: implementation verification, interviews, facility observations, and the judgment calls only experienced professionals can make.
RA/QA teams can run their own document analyses, and auditors working in an external service capacity can use it too.
How the workflow works, step by step
Here’s what the AICA workflow looks like in practice. (You can also watch a short video demo at the top of this explainer or on YouTube to see it all in motion.)
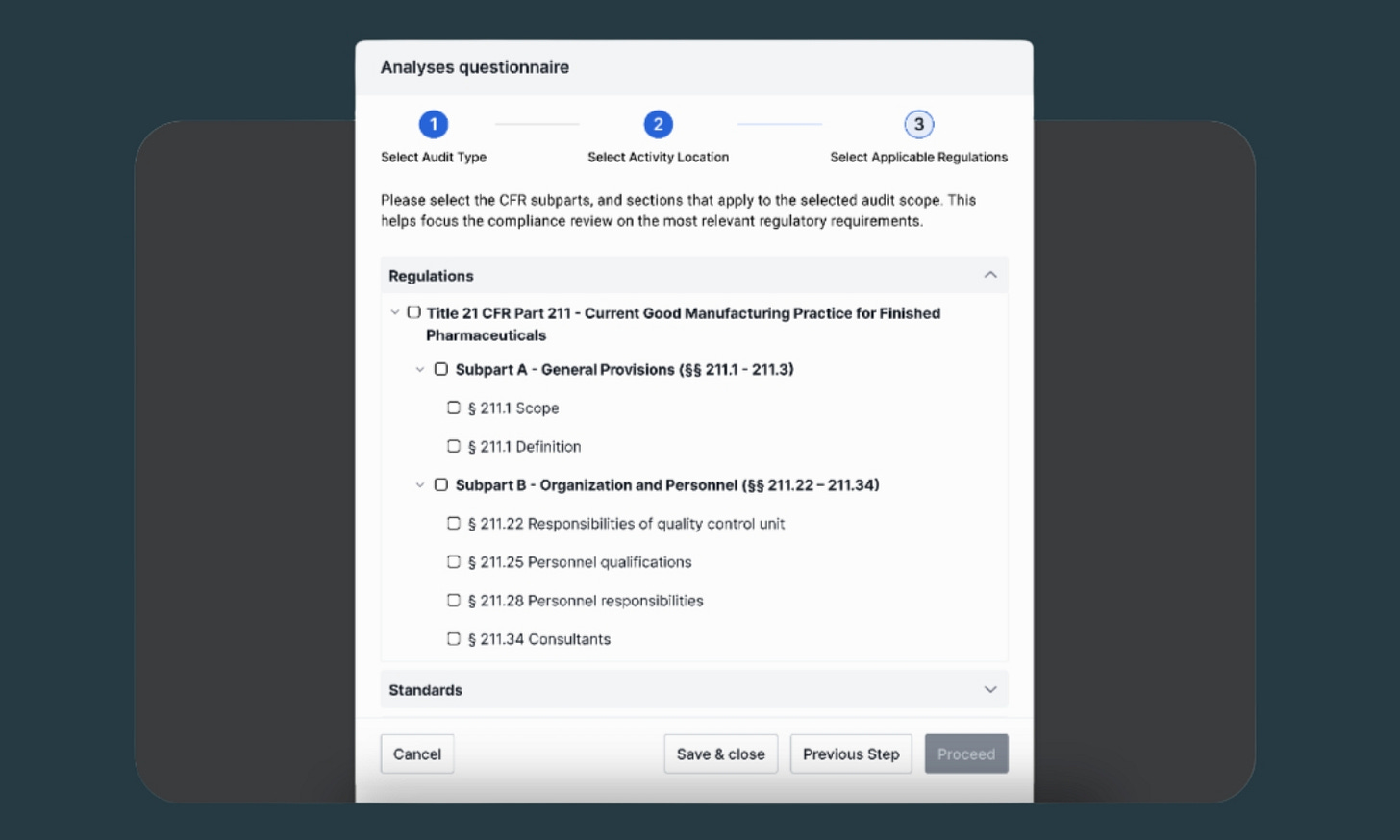
1. Choose exactly which regulations to audit against
Each audit you run in AICA can be customized by selecting the specific regulatory requirements you want to run your documentation against. It’s a simple selection interface: just pick what’s relevant.
There are some interesting use cases here beyond just running routine audits against the same regulations. If you’re planning to expand into a new market, for example, you can run a gap analysis against new requirements well ahead of any official regulatory submission. Preparing for an FDA inspection focused on Part 11? Audit just those requirements. AICA adapts to your compliance priorities, whatever they may be.
AICA currently supports auditing against 21 CFR Part 11 (Electronic Records and Electronic Signatures), 21 CFR Part 211 (cGMP for Finished Pharmaceuticals), and 21 CFR Part 600 (Biological Products). Each is mapped at the section and subsection level so you can customize the scope of every audit.
Additional regulations are in active development, including 21 CFR Parts 50, 54, 56, 58, 312, 314, and 320, along with ICH guidance documents.
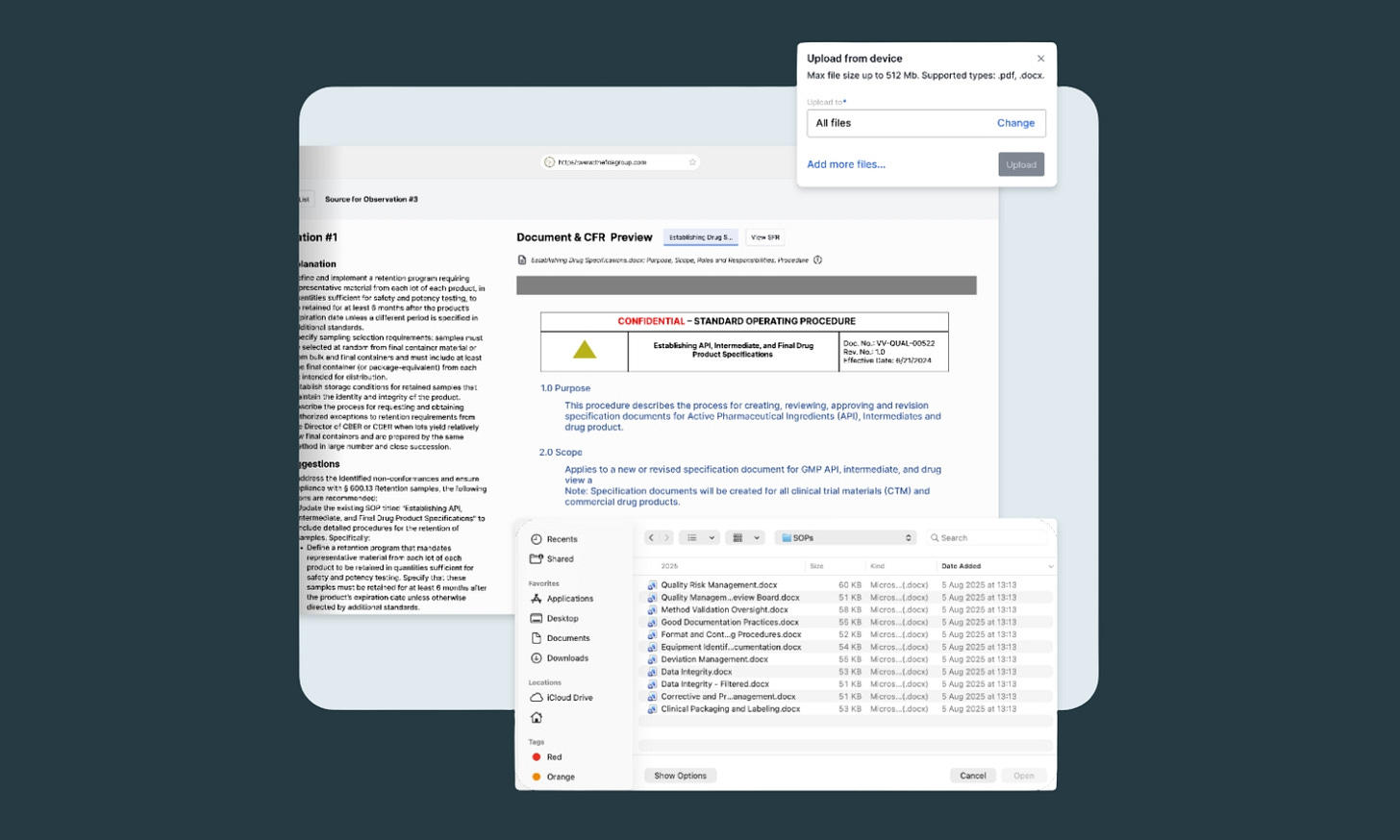
2. Upload and process your QMS documentation in hours
Once AICA knows what you want to audit against, you simply upload your documents for analysis and run it. AICA can process thousands of pages of documentation. What used to require pulling employees off projects for simple document reviews now happens at machine speed.
You can upload documents individually or in batches.
AICA accepts PDF files (including scanned documents via OCR) and Microsoft Word documents (.doc and .docx), which covers the vast majority of QMS documentation in the life sciences industry.
The maximum file size per document is 512 MB (plenty for just about every QMS document).
The platform can handle everything from small, focused audits of 10–20 documents to comprehensive enterprise-wide assessments spanning thousands of documents across your entire QMS. You can also run partial audits on specific subsets of documents (it doesn’t have to be your entire QMS every time, though we recommend comprehensive runs for the most accurate insights).
AICA flags non-conformances, suggests remediation strategies, and generates prioritized action lists. You’ll finally be able to take a meaningful step toward actually proactive compliance monitoring across your QMS.
3. Review observations with your quality team
This is where AICA’s “human in the loop” design comes in. Every observation generated by the AI must be reviewed by a qualified quality professional before the report is finalized. You can include, exclude, or edit any finding based on your institutional knowledge and specific organizational context.
During the review process, you can also add your own observations based on institutional knowledge that the AI might not capture, edit AI-generated observations for clarity or context, or reject observations that don’t apply to your specific situation.
The platform is designed to augment expert judgment, not replace it.
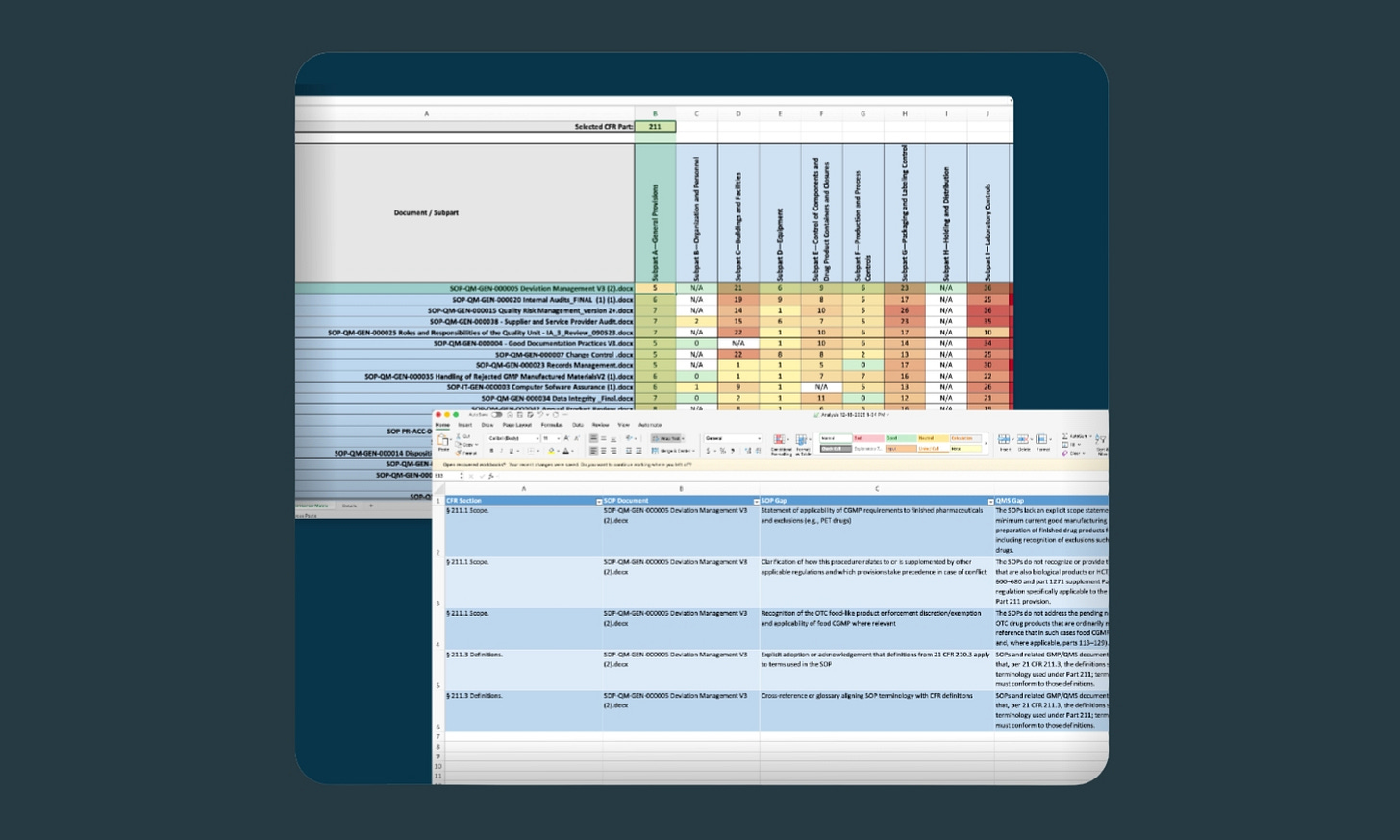
4. Export reports ready for regulatory inspection
After AICA runs its analysis, you get very useful output. Every AICA analysis generates a detailed compliance report with gap analysis, specific CFR references, prioritized observations, and executive dashboards with heat maps for visibility across the entire quality management system.
The idea behind all of this reporting is to support internal audits, supplier audits, and actual regulatory inspections. Each finding that AICA flags includes the specific regulatory clause, the non-conforming document, a clear explanation of the gap, and detailed corrective action recommendations.
Analysis reports remain in your secure AICA workspace until you choose to delete them. These reports do not contain your actual QMS documents — only references to them, along with the gap analysis and recommendations. You can export reports at any time and manage them according to your internal document control procedures.
What makes AICA different from other AI tools?
Apart from what AICA does, one of the most important things to understand about AICA is who built it and how. This isn’t some tech company’s AI that was pointed at regulatory text and told to find problems. This wasn’t vibe-coded in an afternoon by someone looking for a side hustle.
AICA was trained by more than 15 experienced FDA and ISO auditors — people who have actually conducted compliance audits in pharma and biologic companies — using real QMS documentation. The AI model is governed by rules that reflect how experienced auditors actually think about compliance: the practical nuances, the common gaps, the patterns that only come from years of doing this work.
When regulations contain ambiguous language or require professional judgment, AICA provides observations that reflect realistic regulatory expectations based on this expert training. For highly complex, situation-specific requirements, the platform may flag areas for human review rather than attempting to provide definitive assessments.
Stepping back, AICA is unique from similar solutions hitting the market right now in five core ways:
It’s built from the highest-quality audit work. We trained AICA on the depth, rigor, and repeatable patterns behind our most thorough audits — not surface-level checklists or generic compliance rules. Having run thousands of audits and worked with thousands of seasoned auditors, we’ve incorporated all of this directly into the tool’s training, and will continue to train it continuously.
It gives you investigator-level scrutiny at machine speed. AICA reviews large volumes of documentation with the same discipline, judgment, and consistency applied by our audit teams, delivered at the scale AI now unlocks.
You get high-quality analyses in hours, not weeks. Teams can initiate and complete audit-grade analyses without waiting for traditional audit timelines or external scheduling constraints.
You get clear, actionable findings — no rework required. AICA produces structured, decision-ready outputs aligned to our audit methodology, minimizing interpretation gaps and follow-up cycles.
It requires less effort and delivers higher audit quality. AICA reduces the manual review burden while raising the overall depth and consistency of audit outcomes, allowing you to do more with fewer internal resources.
The partnership behind AICA
We built AICA in partnership with EPAM Systems, an NYSE-listed global technology company with a super-impressive track record of life science builds, like:
AICA is built on EPAM’s DIAL open-source GenAI enterprise platform, the same foundation EPAM uses for enterprise AI deployments across industries. This gives AICA a mature, battle-tested infrastructure rather than a custom-built stack we’d have to maintain from scratch.
As Preston Keller, Principal of Life Sciences Business Consulting at EPAM, put it:
“By combining EPAM’s advanced data science and engineering capabilities with The FDA Group’s deep regulatory expertise, we’ve built a platform that addresses a real operational challenge for life sciences companies of all sizes.”
With AICA, EPAM brought the engineering and software firepower while we brought the regulatory expertise.
Security and data privacy in detail
We know this will be one of the first things IT, quality, and procurement teams ask about. Here’s the full picture in clear terms:
Where your data lives: AICA is hosted on Microsoft Azure servers located in the United States. All application databases and storage services are hosted in the Azure region selected for the deployment, ensuring that your information remains physically within U.S. data centers. Azure does not move data outside the country unless explicitly configured for cross-region replication.
How your data is protected: AICA uses enterprise-grade encryption for all data transmissions. Your confidential documents are stored in isolated, access-controlled environments with role-based access controls (RBAC). All access is encrypted, logged, and fully auditable within the secure Azure environment.
Your data is totally private: Your audits and uploaded documents are not viewed by humans. A very small number of authorized employees may access data only when needed for addressing platform security issues or resolving technical problems you report. This access is tightly controlled, audited, and restricted under strict privacy and security policies.
You control document retention: Documents can be deleted immediately after AICA completes its analysis if you prefer not to store them in the platform. You can also configure automated deletion policies based on your organization’s data retention requirements.
On-premises deployment is available: For organizations with stringent security requirements, AICA can be deployed on your own private cloud infrastructure, ensuring your documents never leave your controlled environment.
Implementation: what getting started actually looks like
We’ve designed AICA’s implementation process to be straightforward. We have a whole explainer going into this if you need to circulate it internally — grab the PDF here.
Here’s what the proven process looks like from first conversation to running your first audit:
Introductory Conversation: We start with a needs assessment: understanding your pain points, your current audit process, your regulatory landscape, and your desired outcomes. This is also where we’ll walk you through a live demo of the platform.
Proposal Generation: Based on what we learn, we provide a clear picture of AICA’s capabilities and benefits as they apply to your organization, along with solution options, pricing, and an implementation overview. We’ll also discuss IT security requirements at this stage.
Agreement Processing: This includes the contractual agreement, supplier qualification questionnaire (if your organization requires one), security and compliance review, and confirming timeline and access.
Implementation and Onboarding: This is where you go live. We provide white-glove implementation setup, training and onboarding for your team, a features and workflow orientation, and our total quality guarantee. Typical onboarding takes 1–2 weeks from contract to first audit.
Analyze and Report: Once you’re live, you’ll have a dedicated customer success manager, access to our technical support desk, 24/7 ticketing with a human escalation path, and ongoing compliance reviews and heat mapping.
Setting up AICA itself is simple: once you have a license, we send a welcome email with everything you need to log in and access the platform through your web browser. No additional setup is required. Just upload the documents you want analyzed and begin.
No specialized technical expertise is required. If you can upload documents to a cloud platform (similar to Dropbox or Google Drive), review audit reports and compliance findings, and navigate a web-based interface, you have all the technical skills needed. The platform was designed by regulatory professionals for regulatory professionals.
AICA is optimized for desktop and laptop computers on modern browsers (Chrome, Edge, and Safari are fully supported). Multiple users can access AICA simultaneously, with role-based permissions defined by your organization’s admin during onboarding.
Our approach to pricing
AICA uses a straightforward pricing model that combines an annual platform subscription with a usage-based component based on the volume of documentation you analyze. Pricing is tiered by company size, so smaller organizations aren’t paying an enterprise price tag.
The model was specifically designed to be predictable for pharma budgeting — no surprise bills, no opaque AI compute charges. We walk through the specifics during a demo conversation so we can tailor pricing to your actual QMS size and regulatory needs.
We’ve kept this custom and white-glove for a reason: every organization’s compliance program is different, and we want to make sure the pricing reflects your actual usage, not a one-size-fits-all number.
Teams that reach out during this initial launch period receive preferred pricing.
What’s on the roadmap
AICA is launching today with strong foundational capabilities, and we have an active development roadmap:
More regulations: 21 CFR Parts 50, 54, 56, 58, 312, 314, and 320 are in active development, along with ICH guidance documents. We add new regulations according to a set schedule based on our determination of the most relevant regulations for our customers. If you’re interested in regulations not currently listed, talk to us — your input directly shapes our priorities.
eQMS integrations: Integration capabilities with platforms like MasterControl, Veeva Vault, TrackWise, and others are in development. The goal is to allow AICA to automatically pull the latest versions of your documents for analysis, ensuring you’re always auditing against current documentation. Right now document upload is manual, but your feedback on integration priorities helps us determine which platforms to prioritize.
Additional file formats and language support: AICA currently supports English-language PDF and Word documents. Additional format and language support may be added based on customer feedback and demand.
Continuous AI improvement: The FDA Group regularly reviews all applicable regulations for additions and changes. Updates are first tested internally and once validated, pushed to the current version of AICA. These updates happen at least annually, though they may be more frequent. Customers are notified of updates along with release notes on how the changes impact functionality.
See it for yourself
AICA is available now! The website is live, the platform is ready, and we’re scheduling demos. Visit aica.thefdagroup.com to learn more, explore the platform, and request a demo or ask us anything.
If you’re an Insider subscriber who replied “interested” last week after our sneak peek, we’re already in the process of reaching out. Thank you for being first in line. If you’re getting this via email, feel free to reply to it to let us know you’re interested or head right to our contact form.
We built this because we believe auditing needs an upgrade, and regulatory professionals deserve tools built by regulatory professionals. After more than two decades of deploying expert auditors to the world’s leading life sciences companies, we’ve taken everything we know about what great audits look like and put it into a platform that amplifies your team’s capabilities.
We’re excited to finally share it with you. Talk to us to get the conversation started.
Who is The FDA Group?
The FDA Group helps life science organizations rapidly access the industry's best consultants, contractors, and candidates. Our resources assist in every stage of the product lifecycle, from clinical development to commercialization, with a focus on staff augmentation, auditing, remediation, QMS, and other specialized project work in Quality Assurance, Regulatory Affairs, and Clinical Operations.
With over 3,750 resources worldwide, over 325 of whom are former FDA, we meet your precise resourcing needs through a fast, convenient talent selection process supported by a Total Quality Guarantee.
Here’s why 17 of the top 20 life science firms access their consulting and contractor talent through us:
Resources in 75 countries and 48 states.
26 hours average time to present a consultant or candidate.
Exclusive life science focus and expertise.
Dedicated account management team.
Right resource, first time (95% success).
97% client satisfaction rating.
Talk to us when you're ready for a better talent resourcing experience and the peace of mind that comes with a partner whose commitment to quality and integrity reflects your own.