Why Embedding a Senior Quality Consultant Can Be the Fastest Way to Regain Control
When you have a backlog growing faster than your team can close it, the answer isn't always hiring. Sometimes it's deploying the right person to embed and fix it, fast.
We’re currently working on a new client case study that charts the path of a consultant we embedded in a pharma company for over a year to address a backlog of deviations and the problems that allowed it to emerge in the first place.
Long story short, a large pharma was scaling production of a high-demand drug and found that deviations were outpacing the internal team’s capacity to close them. We embedded a senior deviation specialist on-site, focused on the most complex major deviations requiring thorough root cause analysis.
Over a year, he cleared the critical backlog, became the de facto trainer for other staff on the deviation program, and outlasted multiple different managers at the site. By the time he wrapped up, the system was running smoothly, and the site had regained control.
While we work on telling that story, we first want to talk about this model in general, since it’s worked so well for situations like these across hundreds of firms.
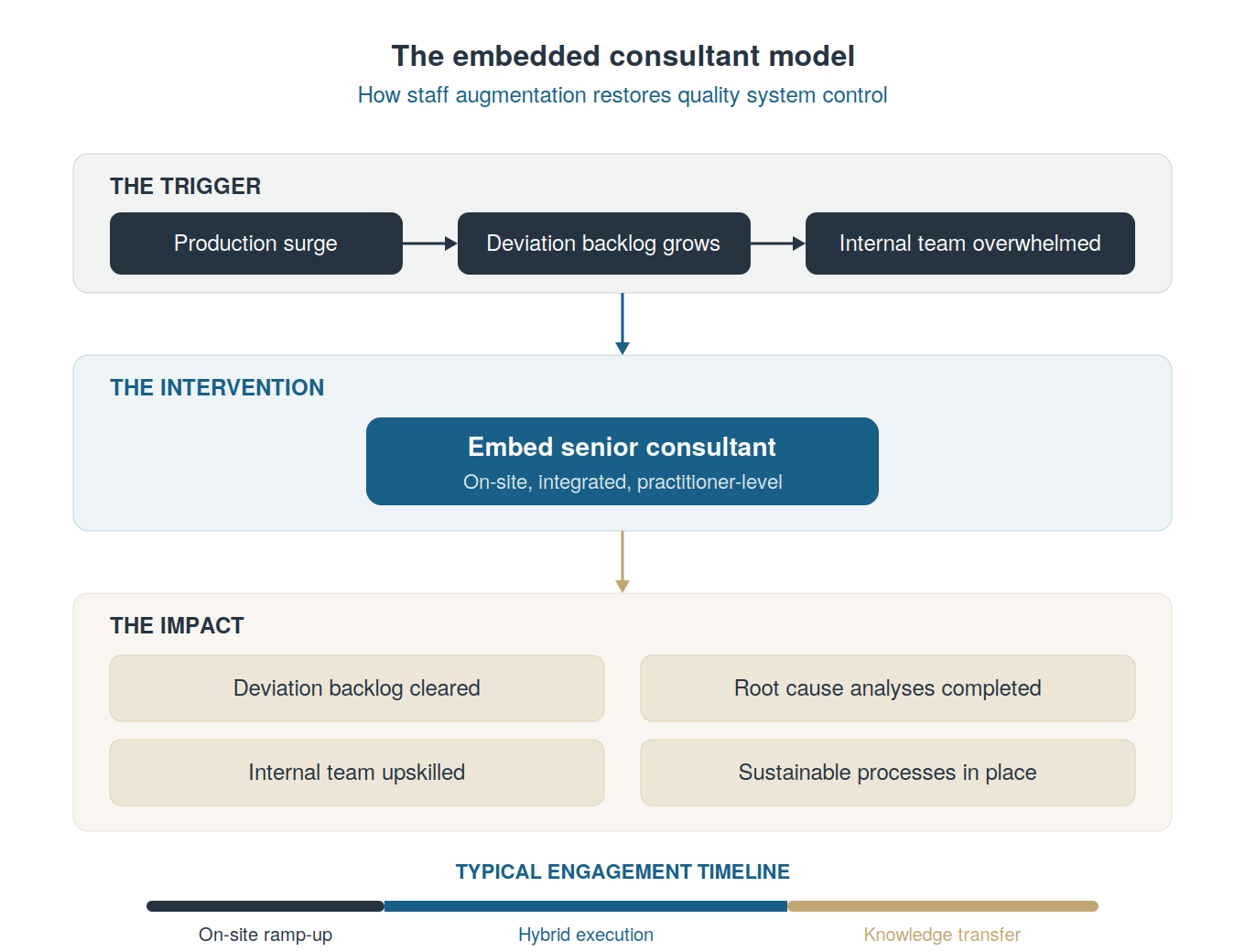
There’s typically a pattern we see over and over in pharma and device manufacturing:
A product goes to market.
Demand surges.
Production scales up.
And then the quality system starts to buckle. Not because anything is fundamentally broken, but because the sheer volume of manufacturing activity generates more deviations, more CAPAs, more investigations than the existing team was ever sized to handle.
A backlog quietly grows and fires start: Major deviations sit open longer than they should, while root cause analyses get delayed. Regulatory risk compounds in the background. And the Quality leadership team, already stretched managing day-to-day operations, doesn’t have the bandwidth or frontline people-power to dig out.
We’ve now supported dozens of teams through exactly this scenario. And one of the most effective interventions we’ve seen (arguably the fastest path back to control) is embedding a senior Quality consultant directly into the operation through staff augmentation (a full-time contractor).
This is not a team of auditors parachuting in for a week, nor is it a gap assessment with a slide deck. It’s a single, highly skilled practitioner or small team who show up, integrate into your team, and start closing deviations on day one.
What "embedding" a consultant actually looks like
Staff augmentation isn’t new in the life sciences, but the mechanics of how it works in practice are worth spelling out because the model is different from typical project-based consulting.
When we embed a consultant, they functionally become part of the client’s RA/QA organization.
They report to an on-site manager who has contact with one of our project managers.
They follow the SOPs.
They attend the team’s meetings and use the systems.
From the outside, they’re generally indistinguishable from an internal employee, except they bring a depth of specialized experience that’s extremely hard to recruit for on a permanent basis.
The engagement typically starts with a full-time on-site presence. In manufacturing environments, that matters! When you’re investigating a deviation tied to a batch record issue or an equipment failure, being able to walk down to the production floor, talk to the operators, and see the process firsthand is very different than trading emails over the course of a week thousands of miles away.
As the consultant builds relationships and becomes fluent in the site’s systems, the arrangement might shift to a hybrid model, depending on the work: a few days on-site, a few days remote, which keeps institutional knowledge flowing while giving the consultant focused time for investigation, writing and root-cause work.
Our contract structure is straightforward: an initial engagement period of typically three to six months, with extensions as the scope of work evolves. In our experience, extensions are the norm rather than the exception.
Once teams see the impact of having an experienced specialist embedded in their operation, the conversation shifts from “when does this end?” to “how do we keep this going?” There’s almost always more work to do that lends itself to having an outside expert working inside the department.
A few other use case examples
While deviation backlogs are the most common trigger, the same approach applies across a range of Quality challenges. Here are a few examples from our recent project history:
Building a Quality department from scratch. A mid-size pharma company with a multi-subsidiary structure needed to stand up an entire Quality function across multiple business units. We started with one Quality lead and scaled to three embedded consultants covering QA, clinical support, supplier qualification, QMS implementation, and training. The team functioned as the company’s Quality department for over three years with zero turnover, adapting as subsidiaries were acquired or divested.
Filling a sudden QA leadership gap. A small biopharma lost its QA manager abruptly. Vendor management was incomplete, SOPs were missing, and there was no formal training system. We deployed a senior QA consultant who, within weeks, stood up a vendor qualification program, began revising SOPs, and built the company’s first electronic training system. Five months later, she transferred knowledge to the permanent hire and stepped out with an overlap period that ensured continuity.
Large-scale backlog remediation with a deployed team. When the backlog is big enough, we deploy teams! One pharma had roughly 150 open deviation investigations growing at 20 per day, with recurring deviations compounding because the backlog was preventing the CAPAs needed to address root causes. We put 17 resources on it: 10 for investigations, 7 dedicated to CAPAs, because clearing deviations without resourcing the downstream CAPA work just moves the bottleneck. The team was delivering results by week two and followed a structured methodology: sample analysis, categorization by expertise, risk-based prioritization, and weekly milestone tracking.
Different scales, different triggers — same underlying model: deploy experienced practitioners who integrate into the operation and do the work while the internal team keeps the day-to-day running.
Why this works so well for so many teams
There are a few reasons the embedded model punches above its weight, and most of them, we’ve come to learn, have nothing really to do with the technical work itself.
Here’s what we’ve seen and heard, having done this for years now:
Speed to impact. A senior consultant who has spent years working on deviations across multiple manufacturing environments doesn’t need a six-month ramp-up period. They’ve seen the patterns before! Batch record errors, bioprocessing deviations, equipment-related issues, process failures. They know what a good root cause analysis looks like. They know what regulators expect to see in a closed investigation. They can start contributing meaningful work almost immediately.
Mentorship as a side effect. This is the benefit that rarely shows up in a statement of work but consistently shows up in the feedback we get when an engagement wraps. When you put a seasoned practitioner alongside less experienced Quality staff, knowledge transfer happens organically. We’ve seen consultants become de facto trainers and mentors for junior team members and newer managers — not because anyone asked them to, but because that’s what good practitioners do. They coach as they work and explain the “why” behind the investigation approach. They model what thorough documentation looks like. For organizations where rapid growth has outpaced internal development, this informal mentorship can be as valuable as the deviation closures themselves.
Continuity through “organizational turbulence.” Fast-growing companies are, almost by definition, volatile. Reporting structures change, managers rotate, layoffs and restructurings happen, and internal teams get reshuffled. Through all of that, an embedded consultant can be a refreshingly stabilizing presence. You have someone there for a few months who maintains institutional knowledge of open investigations and keeps the deviation program moving forward even when leadership is in flux. We’ve had consultants outlast three or four different managers at the same client and still keep their work on track, because their mandate is tied to the work itself, not to any one internal sponsor.
Flexibility that matches the work. Manufacturing deviation backlogs don’t typically shrink on a nicely predictable timeline. As soon as you clear five major deviations, three new ones might land on your desk. An embedded consultant model accommodates this reality in a way that fixed-scope project engagements can’t. Extensions are simple; the scope can flex. And when the backlog is finally under control (when the system is running smoothly and the team can sustain it internally), the engagement winds down naturally. (In some cases, our clients have offered FTE opportunities to our consultants to keep them on permanently.)
Backlogs (and similar problems) tend to be capacity issues
The vast majority of backlog root causes we’ve identified are genuine capacity problems, which is why the embedded model is so well-suited to addressing them. It almost never has anything to do with the team's competence. Some of the most impressive teams we’ve ever worked with deal with backlogs.
In reality, most Quality teams are sized to handle a specific volume of production. When production ramps dramatically or something else affects the team, the ratio of Quality resources to manufacturing output gets thrown off. Deviations are the downstream symptom.
Hiring permanent staff to close the gap takes months, sometimes the better part of a year, when you factor in recruiting, onboarding, and ramp-up time for roles that require regulatory knowledge and site-specific training. And there’s a legitimate question about whether you even want to permanently expand headcount for what might be a temporary surge.
That’s the sweet spot for staff augmentation: You need experienced people now. You need them integrated into the operation, not advising from the outside. And you need the flexibility to scale the support up or down as conditions change. An embedded consultant (or a small team of them) fills that gap in weeks, not quarters.
What makes a good embedded consultant
Not every consultant or subject matter expert who works on contract is suited for this kind of work. Staff augmentation demands a specific temperament alongside technical expertise.
The best embedded consultants are, first and foremost, practitioners. They’ve done the work themselves. They’ve written the investigations, performed the root cause analyses, and closed the deviations. They’re not just strategists who can solve problems. They’re people who can sit down and do the job, too.
Beyond that, they need to be adaptable. Every site has its own culture, its own SOPs, its own political dynamics. A consultant who can’t read a room, build trust with operators and managers, and adjust their approach to fit the client’s way of working will struggle regardless of how strong their technical skills are.
And frankly, the best people function as what you might call “organizational connective tissue.” They build relationships across departments. They keep their project managers informed. They flag risks and opportunities that go beyond their immediate scope. One of our consultants once
One of our consultants often describes the role using a metaphor about bees carrying pollen from flower to flower, leaving something beneficial at every stop. It’s a funny image, but it captures something real about what happens when you put a well-connected, well-intentioned practitioner inside a complex organization. They bring their best practices with them and deposit them inside the org.
When to consider staff augmentation
If any of the following sound familiar, an embedded senior Quality consultant may be exactly what you need:
Your deviation backlog is growing faster than your team can close it.
You have major deviations that have been open for months without a clear path to closure.
Your Quality team is spending so much time on day-to-day firefighting that proactive work has stalled.
You’ve recently scaled production significantly, and your Quality infrastructure hasn’t caught up.
You’ve been through a reorganization or layoffs and lost institutional knowledge in the Quality function.
These are the situations we get called into regularly. And the embedded model is consistently one of the most effective and efficient ways to regain control. If you’re dealing with a Quality capacity challenge and want to explore whether staff augmentation is the right fit, learn more about how we use the model and get the conversation started.
Who is The FDA Group?
The FDA Group helps life science organizations rapidly access the industry's best consultants, contractors, and candidates. Our resources assist in every stage of the product lifecycle, from clinical development to commercialization, with a focus on Quality Assurance, Regulatory Affairs, and Clinical Operations.
With thousands of resources worldwide, hundreds of whom are former FDA, we meet your precise resourcing needs through a fast, convenient talent selection process supported by a Total Quality Guarantee. Learn more and schedule a call with us to see if we’re a fit to help you access specialized professionals and execute your projects on time and on budget.